Conserved interdomain linker promotes phase separation of the multivalent adaptor protein Nck. - Abstract - Europe PMC

PDF) Nucleocytoplasmic Shuttling of the Adapter Protein SH2B1β (SH2-Bβ) Is Required for Nerve Growth Factor (NGF)-Dependent Neurite Outgrowth and Enhancement of Expression of a Subset of NGF-Responsive Genes

Kinase inhibition profiles as a tool to identify kinases for specific phosphorylation sites | Nature Communications

PDF) Selectivity and promiscuity in the interaction network mediated by protein recognition modules | Luisa Castagnoli and Serena Paoluzi - Academia.edu
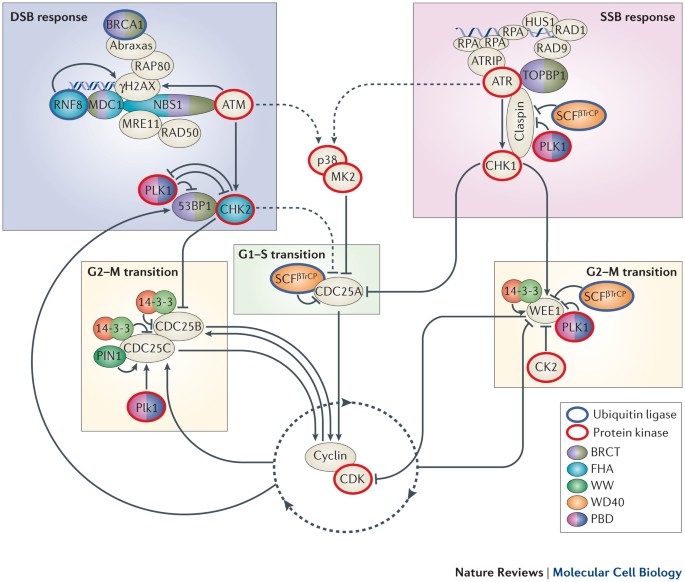
Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response | Nature Reviews Molecular Cell Biology
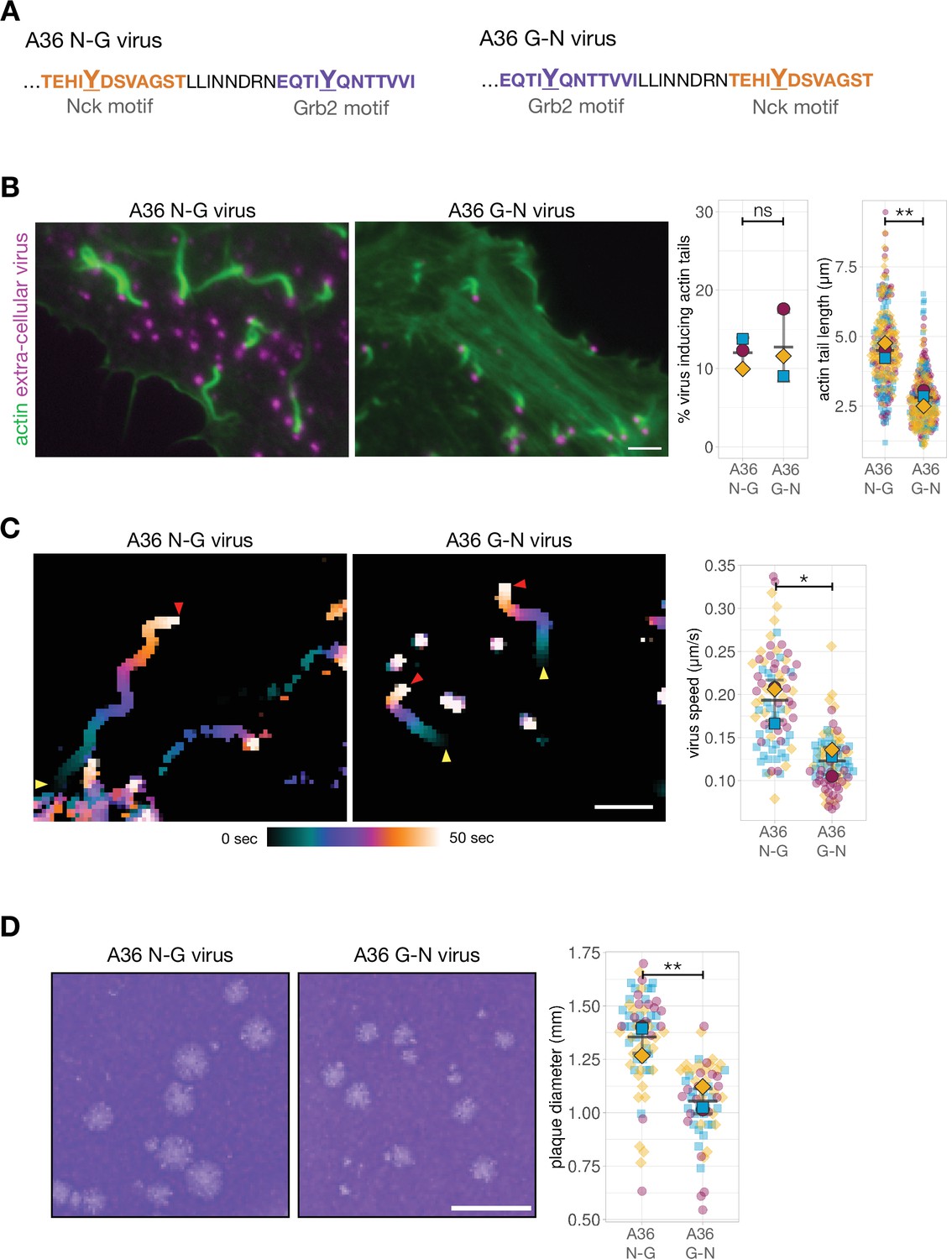
The relative binding position of Nck and Grb2 adaptors impacts actin-based motility of Vaccinia virus | eLife

KINATEST-ID: A Pipeline To Develop Phosphorylation-Dependent Terbium Sensitizing Kinase Assays | Journal of the American Chemical Society

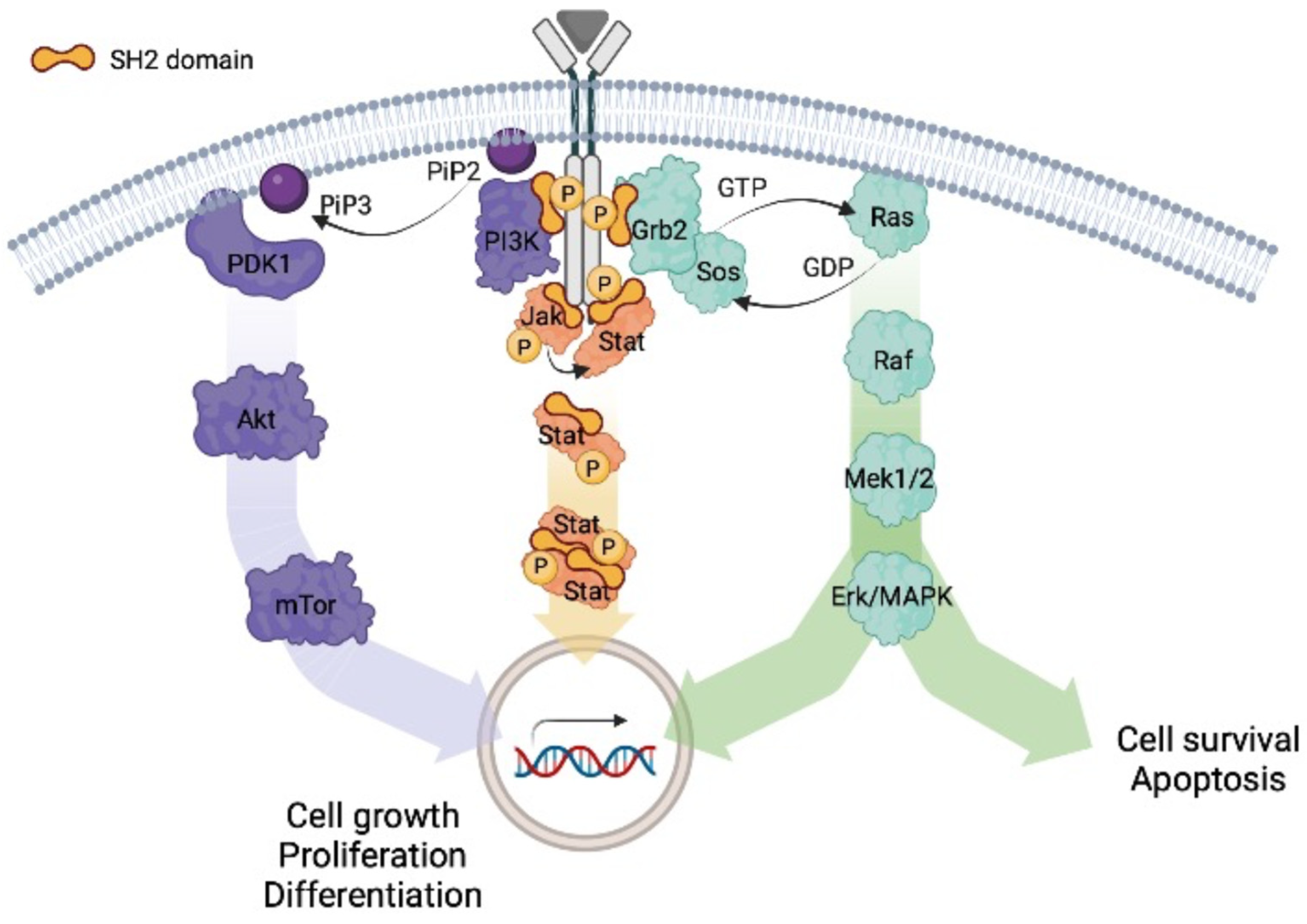
A structural perspective on targeting the RTK/Ras/MAP kinase pathway in cancer - Heppner - 2021 - Protein Science - Wiley Online Library

Conserved interdomain linker promotes phase separation of the multivalent adaptor protein Nck | PNAS

A Methodological Assessment and Characterization of Genetically-Driven Variation in Three Human Phosphoproteomes | Scientific Reports
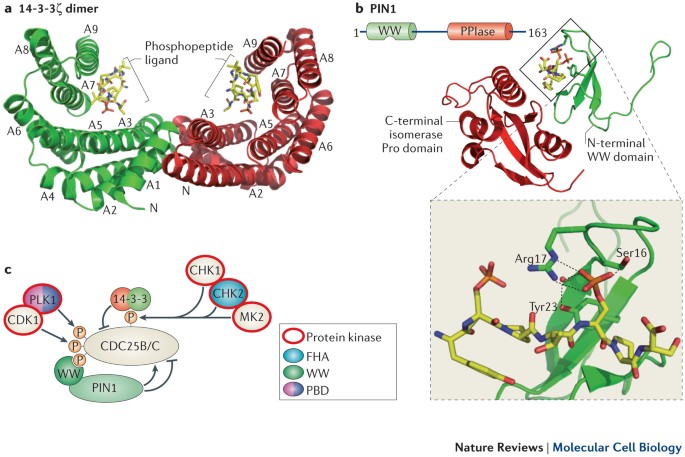
Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response | Nature Reviews Molecular Cell Biology

RNA Polymerase II C-Terminal Domain: Tethering Transcription to Transcript and Template | Chemical Reviews

Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response | Nature Reviews Molecular Cell Biology